At Anthony Nolan, over a third of our 300 staff are scientists. Our research team is always taking on groundbreaking new initiatives to improve transplantation and quality of life for patients. And that’s why we asked one of our PhD-student researchers, Vikesh Devlia, to write our latest blog post and give you a glimpse into their lifesaving work.
In this article, the first in a series of blogs looking at our science and research, Vikesh explains about the new research project he’s working on; a study of how long it takes the immune system to regenerate in patients who’ve had a cord blood transplant.
Why umbilical cord blood matters
Patients with blood cancers or blood disorders can be treated using a variety of stem cell sources, such as bone marrow or peripheral blood, in an attempt to cure their disease – but in the last 20 years, stem cells from umbilical cord blood have become an increasingly popular option.
However, patients can suffer from complications post-transplant, and they’re often prone to infection due to the naivety of their immune system.
They can even experience Graft versus Host Disease (GvHD), a complication that arises post-transplant when the transplanted donor cells begin to attack the recipient’s body. [read more about GvHD on our website here]
Our IRES study
To address this problem, we set up the immune reconstitution (IRES) study in 2009 to identify how long a patient’s immune system takes to fully redevelop after a transplant that uses umbilical cord blood.
We wanted to identify this time-frame so we could tailor post-transplant protocols, manage outcomes like infection and GvHD more effectively, and limit complications.
From 2011 onwards, we began recruiting umbilical cord blood transplanted patients from 13 hospitals across the UK, with informed consent.
We collect blood samples from patients at six different times – 28, 60, 100, 180, 365 and 720 days post-transplant.
The blood samples are then sent back to the Anthony Nolan Research Institute and used in two main processes:
- Firstly, we identify the number of specific immune cells, by staining a small volume of the sample with fluorescent antibodies and feeding them into a machine called a flow cytometer.
- Then we separate the blood and store the white blood cells away for future work. This is so further genetic work and research work can be undertaken from the same patient sample.
I’ve been working on the study since 2012, collecting information from transplant centres about patients who’ve been recruited at the respective sites.
Once the patients have consented, I send out a blood sample kit. The nurses at the transplant center then collect blood samples from the patients and the kits are sent back to me to be processed as described.
The day-to-day tasks for the study vary; samples are sent to us frequently and must be processed right away to ensure that we achieve the best results! Afterwards, we analyse the samples, generating data which is then entered onto a bespoke database.
Database management, too, must be undertaken frequently to ensure that the data’s ready for analysis once all of the patient samples have been collected.
Our results
So far, we’ve recruited 80 patients onto the study and we’ll be continuing to recruit them until July 2016. In the meantime, we’re working on the research data, which will then be compared and linked to clinical data, in order to understand the outcomes that occur within patients post-transplant.
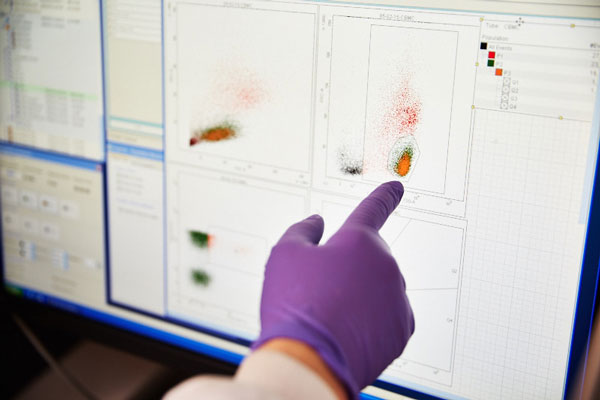
Using this information, the aim is to produce a timeline which will help clinicians to tailor post-transplant regimens, so that future cord blood transplanted patients are at a reduced risk of infection and GvHD development.
I’m proud of the study, and I think it’s making excellent progress – we’re the first group within the UK to study this cohort of patients with such a broad range of cells to identify.
At the end of it all, there’ll be a large dataset generated, and the clinical data will help us to understand patient outcomes better than ever before.
